The FDA, FTC and Cosmetics
In 1938, the American Federal Government passed two laws that would have a dramatic effect on the operations of the American cosmetic industry – The Wheeler-Lea Act (March 21, 1938) and the Food, Drug, and Cosmetic Act (June 25, 1938).
Food, Drug, and Cosmetic Act
The Food, Drug, and Cosmetic Act (FD&CA), which replaced the Federal Pure Food and Drug Act of 1906, improved the ability of the Food and Drug Administration (FDA) to control dangerous or deceptive food and drugs and extended its responsibilities to include medical devices and cosmetics. The Act’s long title set out its main aims: “To prohibit the movement in interstate commerce of adulterated and misbranded food, drugs, devices, and cosmetics, and for other purposes.”
The FDA had been highlighting deficiencies in 1906 Act for some time. In 1933, two FDA staff members – its chief education officer, Ruth deForest Lamb [1896-1978] and chief inspector, George Larrick [1901-1968] – had set up a travelling exhibition that displayed a range of worthless, dangerous or deceptive products that were legal under the existing law.

Above: Food and Drug Administration travelling exhibition.
Amongst other items in the exhibition were some dangerous cosmetics which, along with therapeutic devices, were not covered by the 1906 Act. These cosmetics included: Lash Lure, an eyebrow and eyelash dye involved in a case where a woman had been blinded; hair dyes that contained lead; and Koremlu, a hair remover made with thallium acetate, a dangerous poison.
See also: Lash Lure and Chemical Depilatories
Cosmetics
The sections of the FD&CA that dealt with cosmetics were designed to ensure that cosmetics and perfumes did not contain harmful ingredients; were not placed in deceptive packaging; were labelled according to a set of specifications laid down by the Act; and were not subject to false, misleading or unwarranted claims.
Harmful ingredients
As well as the products already mention there were a few additional cosmetics the FDA considered to be potentially harmful and the agency seized a number of products after the Act was passed.
The FD&CA also placed strict controls on the manufacture and use of cold-tar dyes. Approved dyes were classified according to whether they could be used in foods, drugs and cosmetics (FD&C), drugs and cosmetics (D&C) or for externally applied drugs and cosmetics (ext. D&C). This required some cosmetics to be reformulated to remove colours that were not sanctioned for use in cosmetics.
Drugs were considered to be potentially more harmful than cosmetics and were more tightly controlled. The FD&CA defined a drug as any product that “intended [my italics] to affect the structure or any function of the body of man or other animals”. The FDA rightly considered ingredients when determining whether a product should be classified as a drug but also took product claims into account. This meant that a product normally regarded as a cosmetic, could be classified as a drug within the meaning of the Act if therapeutic effects were made for it. For example, hair preparations that only said they would beautify the hair were considered to be cosmetics but those that professed to be useful in the treatment of dandruff or to stop hair loss would be classified as drugs. American cosmetic firms had to be careful about their product claims to avoid more onerous restrictions.
Labelling and packaging
The FD&CA introduced new labelling standards for cosmetics. Packages now had to included the name of the product, the name of the manufacturer, packer, or distributor as well as the quantity of product the pack contained.
Deceptive and slack-filled packaging was also prohibited under the terms of the Act. Noticeable difference between the expectations a consumer got from the packaging and the amount of product it actually contained were also more tightly controlled. This required some manufacturers to repackage and/or relabel their cosmetics, something many had to do again when packaging restrictions began following America’s entry into the Second World War.
False, misleading or unwarranted claims
The FDA’s ability to control false, misleading or unwarranted claims for foods, drugs and cosmetics was severely limited by the terms of the FD&CA. Early drafts of the bill had included controls on advertising but the final form of the Act limited the FDA’s jurisdiction to product labelling, a responsibility it had previously held for food and drugs under the 1906 Act. Control over advertising, where most consumers got their information and where most spurious product claims were made, continued to be the concern of Federal Trade Commission under the terms of the Wheeler-Lea Act.
Wheeler-Lea Act
The Federal Trade Commission (FTC) had been established in 1914 to enforce the Clayton Antitrust Act, Federal legislation designed to prevent anticompetitive practices. This included jurisdiction over the advertising of all commodities that travelled interstate.
The FTC had become increasingly concerned about deceptive advertising. In 1929, it had established a special board of investigation to survey newspaper and magazine advertising for misleading claims and extended this to radio advertising in 1934. However, under the terms of the Clayton Antitrust Act, the FTC could only act against a deceptive advertisement if it could show that the advertisement was likely to cause a competitor to lose business.
The Wheeler-Lea Amendment to the Federal Trade Commission Act gave the FTC authority to prevent false advertising of food, drugs, devices, and cosmetics solely on the basis that an advertisement intended to deceive. This removed the previous limit that it had to harm a competitor, extending protection from false or deceptive advertising to consumers as well as businesses.
Joint responsibility
The Wheeler-Lea Act and Food, Drug, and Cosmetic Act therefore divided responsibility for false, misleading or unwarranted claims between the FDA and the FTC along the lines that had existed before 1938. The FDA was primarily concerned with the safety of cosmetics but also monitored how they were labelled, a responsibility that also extended to any published material accompanying the product such as brochures or pamphlets. The FTC, on the other hand, enforced standards for cosmetic advertising.
To assist the cosmetic industry, the FDA and FTC separately released lists of words and phrases that they no longer expected to see associated with cosmetics, the FDA for product labels, the FTC for advertising. These lists were published in industry journals and, along with books and other publications on the subject, meant manufacturers were well informed about the new restrictions. However, they did not resolve the problem of duplication of control.
As every manufacturer and merchandiser of cosmetics and soaps and flavoring essences knows, this law has placed the policing of all advertising under the supervision of the Federal Trade Commission. But the perplexing phase to the Food and Drug Administration, as well as to the business man, is the fact that the labels on containers or packages must be policed by the Food and Drug people. Even the Food and Drug people regard this system of duplication as almost arrant nonsense. In confidence they will tell you that they sympathize with the confusion of the industry and with the irritation of industry over what seems like unnecessary duplication. To still further complicate matters, the Department of Justice enters the field when actual litigation begins.
(Kruckman, 1940, p. 32)
The division between labelling and advertising remained a source of friction between the FDA and the FTC until 1954 when the two agencies released a joint statement outlining their individual and joint responsibilities.
Cosmetics
To summarise, the Wheeler-Lea Act and the Food, Drug, and Cosmetic Act required cosmetic companies to make packaging changes, abide with the regulations associated with coal-tar dyes, comply with the restrictions on cosmetics that were also classified as drugs, such as hormone creams and antiperspirants, and take more care with the claims made for their products.
It was the need to avoid false, misleading or unwarranted claims that gave cosmetic companies the most trouble. For example, both the FDA and the FTC came down heavily on cosmetics that gave the impression that they had a food value. Statements that a cream nourished, fed, rejuvenated or revitalised the skin, built new skin, banished fatigue, or acted as a tonic for nerves, were all claims considered to be scientifically unfounded. Closely associated with ‘skin foods’ were the ‘muscle oils’ which were supposed to nourish the muscles and make them full and firm once more.
The FDA insisted that labels such as ‘Skin Food’, ‘Tissue Cream’ or ‘Muscle Oil’ were removed from packaging and the FTC clamped down on nutritive advertising claims. For example, Elizabeth Arden, much to her annoyance, was forced to stop advertising that her products ‘furnish nourishment to the skin’ and had to change the name of her Orange Skin Food to Orange Skin Cream and her Muscle Oil to Moisture Oil.
See also: Skin Foods and Muscle Oils
The advertising value of nutritive claims for skin creams appears to have been important to cosmetic companies. Even though they were restricted from making such claims in the United States, American cosmetic companies like Dorothy Gray continued to make nutrient claims for these products overseas.
Consequences
The effect of the Wheeler-Lea Act and the Food, Drug, and Cosmetic Act on the American cosmetic industry was wide ranging. One consequence that may have gone unnoticed was a possible stultifying effect the advertising restrictions had on the sale of skin creams. It may just be a coincidence but there are reports of sales of American skin creams going into decline for the next fifteen years.
Up until a few years ago, and for a period of nearly 15 years before that, cosmetic creams were undergoing a substantial decline, not only in volume but in the number of women using such creams and in the frequency of use. This has been emphatically confirmed by all newspaper, magazine or professional research surveys.
(Drug and Cosmetic Industry, 1959, p. 322)
The decline in the sales may have been a consequence of the limitations placed by the FTC and the FDA on nutritional claims for cosmetics that were widely used in American skin-care advertisements before 1938. It would not be until the 1950s, following the introduction of new biologicals like royal jelly, and the development of the moisturiser, that sales would show signs of improvement.
See also: Moisturisers
First Posted: 30th August 2022
Last Update: 1st October 2023
Sources
Abelson, N. (1940). Checklist of FTC cosmetic taboos. The American Perfumer and Essential Oil Review, March, 41-44, 90.
Control under the Wheeler-Lea Act. (1940). The Drug and Cosmetic Industry, 46(5), May, 548-551.
Food, Drug and Cosmetic Act. (1940). The Drug and Cosmetic Industry, 46(5), May, 538-547.
Krukman, A. (1940). FTC law enforcement changes pending. The American Perfumer and Essential Oil Review, May, 31-32, 88.
MacIntyre, E. (1963). Statement on fair advertising landmarks at a meeting of the food, drug and cosmetic law section of the New York State Bar Association. Retrieved May 26, 2022, from https://www.ftc.gov/system/files/documents/public_statements/683461/19620122_macintyre_fair_advertising_landmarks.pdf
Mill, R. J. (1942). Food, drug and cosmetic act as it relates to cosmetics, regulations and interpretations. In M. G. deNavarre, The chemistry and manufacture of cosmetics (pp. 631-689). Boston, MA: D. Van Nostrand Company, Inc.
Milton H., (1939). The Control of False Advertising Under the Wheeler–Lea Act, 6 Law and Contemporary Problems, Winter, 91-110 Retrieved May 26, 2022, from https://scholarship.law.duke.edu/lcp/vol6/iss1/5

1923 Dr. Royal S. Copeland [1868-1938] physician, public health officer, and United States Senator, a major supporter of the Food, Drug and Cosmetic Act who died just days before it became law.
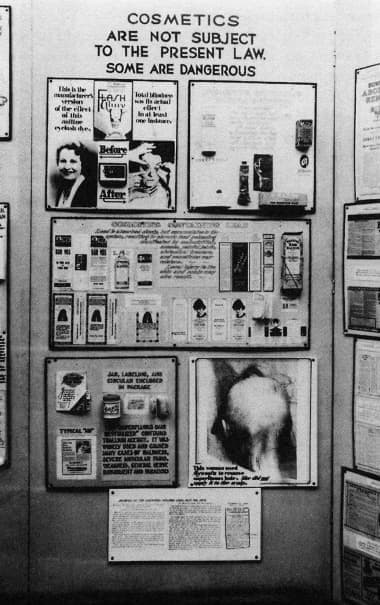
Food and Drug Administration display that includes information on Lash Lure, cosmetics that contain lead, and Koremlu.

Closer view of the Lash Lure display.
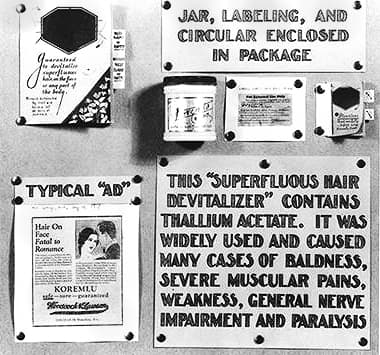
Closer view of the Koremlu display.

1939 Food and Drug Administration. Misleading cosmetic claims (The American Perfumer, September 1939, May 1940, p. 27).

1940 Abelson checklist for drug and cosmetic advertising claims outlawed by the FTC.

1940 Federal Trade Commission. Statements that cannot be made about any cosmetic (Drug & Cosmetic Industry, May 1940, p. 550).

1944 Book on ‘The law of foods, drugs & cosmetics’ by Harry A. Toulmin.

1949 Dorothy Gray ‘Rule of Three’ in America: Cleanse, Stimulate, Lubricate.

c.1954 Dorothy Gray ‘Rule of Three’ in Britain: Cleanse, Stimulate, Nourish.
